|
11/16/2023 0 Comments Elon musk dispositivo cerebral
These devices aim to cure a range of conditions from obesity, autism, depression and schizophrenia, and to enabling web browsing and telepathy. O executivo mostrou o dispositivo funcionando e sendo capaz de reconhecer quando um porco, que tinha o implante, usava o focinho para farejar ou tocar alguma. Musk envisions both disabled and healthy individuals swiftly getting surgical implants at local centers. A neurotech Neuralink, uma empresa do bilionrio Elon Musk, apresentou nesta sexta-feira, 28, atualizaes de um produto parar criar uma ligao direta entre crebros e computadores. He made headlines late last year when he said he was already so confident in the device’s safety that he would be willing to implant them in his own children. Over the years, Musk has publicly outlined an ambitious plan for Neuralink. 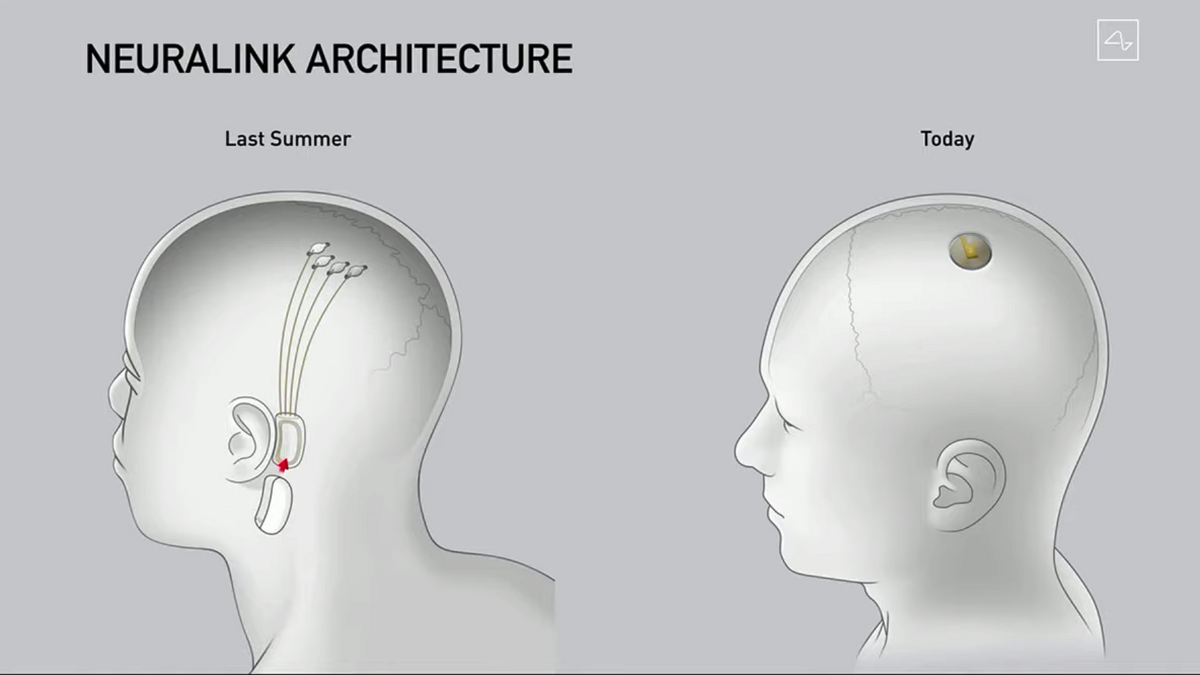
“This is the result of incredible work by the Neuralink team in close collaboration with the FDA and represents an important first step that will one day allow our technology to help many people,” the company said in a tweet on Thursday. In a tweet on Thursday, Neuralink said it was not yet open for a clinical trial. The inquiry has also been looking at the USDA’s oversight of Neuralink. 
The company has killed about 1,500 animals, including more than 280 sheep, pigs and monkeys, following experiments since 2018, Reuters previously reported. Last year, the USDA’s inspector general began investigating, at the request of a federal prosecutor, potential violations of the Animal Welfare Act, which governs how researchers treat and conduct tests on certain types of animals. Neuralink has already been the subject of federal investigations. Em julho de 2019, Elon Musk, CEO da SpaceX e da Tesla, apresentou definitivamente como estava o andamento de um de seus projetos mais recentes: a Neuralink. Thursday’s FDA approval comes as US lawmakers are urging regulators to investigate whether the makeup of a panel overseeing animal testing at Neuralink contributed to botched and rushed experiments. Major issues involved the lithium battery of the device, the possibility of the implant’s wires migrating within the brain and the challenge of safely extracting the device without damaging brain tissue. O processo de instalao do aparelho deve ser semelhante cirurgia ocular a laser Lasik, segundo Musk, com fios e mquina inseridos no paciente por um rob em um procedimento. The FDA had pointed out several concerns to Neuralink that needed to be addressed before sanctioning human trials, according to the employees. A inteno do dispositivo desenvolvido por Elon Musk criar uma uma conexo direta entre um computador e o chip inserido no crebro. Yet the company, founded in 2016, only sought FDA approval in early 2022 – and the agency rejected the application, seven current and former employees told Reuters in March.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
